How to Draw Molecular Orbital Diagram of Co
Chapter 8. Advanced Theories of Covalent Bonding
8.4 Molecular Orbital Theory
Learning Objectives
By the cease of this section, you lot volition exist able to:
- Outline the basic breakthrough-mechanical approach to deriving molecular orbitals from atomic orbitals
- Describe traits of bonding and antibonding molecular orbitals
- Calculate bond orders based on molecular electron configurations
- Write molecular electron configurations for first- and second-row diatomic molecules
- Relate these electron configurations to the molecules' stabilities and magnetic properties
For virtually every covalent molecule that exists, we can now draw the Lewis construction, predict the electron-pair geometry, predict the molecular geometry, and come up close to predicting bond angles. However, one of the nearly important molecules we know, the oxygen molecule Oii, presents a problem with respect to its Lewis structure. We would write the following Lewis structure for O2:
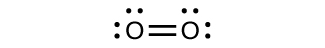
This electronic structure adheres to all the rules governing Lewis theory. In that location is an O=O double bail, and each oxygen atom has viii electrons effectually information technology. However, this picture is at odds with the magnetic beliefs of oxygen. By itself, Otwo is not magnetic, but information technology is attracted to magnetic fields. Thus, when we pour liquid oxygen past a strong magnet, it collects between the poles of the magnet and defies gravity, as in Figure 1 in Chapter 8 Introduction. Such attraction to a magnetic field is called paramagnetism, and it arises in molecules that accept unpaired electrons. And yet, the Lewis structure of O2 indicates that all electrons are paired. How do we account for this discrepancy?
Magnetic susceptibility measures the force experienced by a substance in a magnetic field. When we compare the weight of a sample to the weight measured in a magnetic field (Figure 1), paramagnetic samples that are attracted to the magnet will appear heavier because of the force exerted by the magnetic field. We tin can summate the number of unpaired electrons based on the increase in weight.
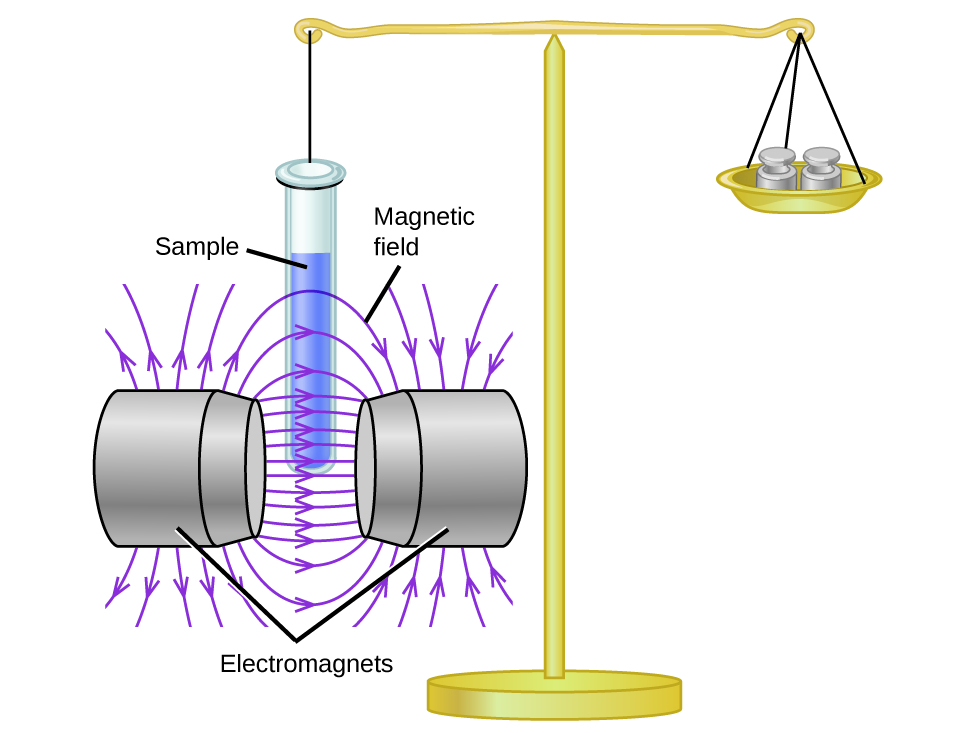
Experiments show that each Otwo molecule has 2 unpaired electrons. The Lewis-structure model does not predict the presence of these two unpaired electrons. Unlike oxygen, the credible weight of most molecules decreases slightly in the presence of an inhomogeneous magnetic field. Materials in which all of the electrons are paired are diamagnetic and weakly repel a magnetic field. Paramagnetic and diamagnetic materials do non act as permanent magnets. Merely in the presence of an applied magnetic field do they demonstrate attraction or repulsion.

Water, like most molecules, contains all paired electrons. Living things comprise a large percentage of h2o, and so they demonstrate diamagnetic behavior. If you place a frog about a sufficiently big magnet, it will levitate. You tin see videos of diamagnetic floating frogs, strawberries, and more.
Molecular orbital theory (MO theory) provides an explanation of chemical bonding that accounts for the paramagnetism of the oxygen molecule. It also explains the bonding in a number of other molecules, such every bit violations of the octet dominion and more molecules with more than complicated bonding (across the scope of this text) that are hard to describe with Lewis structures. Additionally, it provides a model for describing the energies of electrons in a molecule and the probable location of these electrons. Different valence bond theory, which uses hybrid orbitals that are assigned to 1 specific atom, MO theory uses the combination of diminutive orbitals to yield molecular orbitals that are delocalized over the entire molecule rather than existence localized on its constituent atoms. MO theory also helps united states of america understand why some substances are electrical conductors, others are semiconductors, and still others are insulators. Table 2 summarizes the main points of the two complementary bonding theories. Both theories provide different, useful means of describing molecular structure.
| Valence Bond Theory | Molecular Orbital Theory |
|---|---|
| considers bonds every bit localized betwixt i pair of atoms | considers electrons delocalized throughout the entire molecule |
| creates bonds from overlap of atomic orbitals (s, p, d…) and hybrid orbitals (sp, sp 2, sp 3…) | combines atomic orbitals to form molecular orbitals (σ, σ*, π, π*) |
| forms σ or π bonds | creates bonding and antibonding interactions based on which orbitals are filled |
| predicts molecular shape based on the number of regions of electron density | predicts the arrangement of electrons in molecules |
| needs multiple structures to describe resonance | |
| Table 2. Comparison of Bonding Theories | |
Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. Just like electrons around isolated atoms, electrons around atoms in molecules are express to discrete (quantized) energies. The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital (Ψ 2). Similar an atomic orbital, a molecular orbital is total when it contains two electrons with opposite spin.
Nosotros will consider the molecular orbitals in molecules composed of ii identical atoms (H2 or Cl2, for instance). Such molecules are called homonuclear diatomic molecules. In these diatomic molecules, several types of molecular orbitals occur.
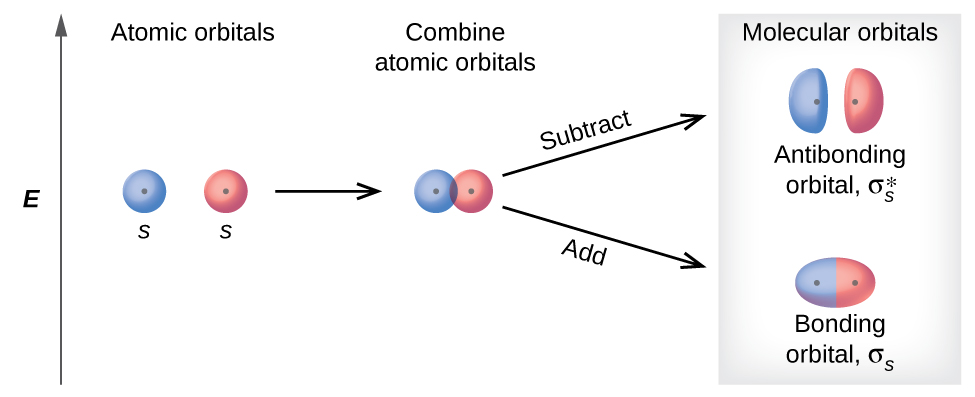
The mathematical process of combining diminutive orbitals to generate molecular orbitals is chosen the linear combination of atomic orbitals (LCAO). The wave function describes the wavelike properties of an electron. Molecular orbitals are combinations of atomic orbital wave functions. Combining waves can lead to constructive interference, in which peaks line up with peaks, or destructive interference, in which peaks line upwards with troughs (Figure 2). In orbitals, the waves are three dimensional, and they combine with in-stage waves producing regions with a higher probability of electron density and out-of-phase waves producing nodes, or regions of no electron density.

There are two types of molecular orbitals that tin can form from the overlap of two atomic s orbitals on adjacent atoms. The two types are illustrated in Figure 3. The in-phase combination produces a lower free energy σ s molecular orbital (read as "sigma-s") in which most of the electron density is directly between the nuclei. The out-of-phase add-on (which tin also be thought of as subtracting the wave functions) produces a higher energy [latex]\pmb \sigma^*_s[/latex]molecular orbital (read every bit "sigma-south-star") molecular orbital in which there is a node between the nuclei. The asterisk signifies that the orbital is an antibonding orbital. Electrons in a σ s orbital are attracted by both nuclei at the aforementioned fourth dimension and are more stable (of lower energy) than they would be in the isolated atoms. Adding electrons to these orbitals creates a forcefulness that holds the 2 nuclei together, so we call these orbitals bonding orbitals. Electrons in the [latex]\sigma^*_s[/latex] orbitals are located well away from the region between the two nuclei. The attractive force between the nuclei and these electrons pulls the 2 nuclei apart. Hence, these orbitals are called antibonding orbitals. Electrons fill up the lower-energy bonding orbital earlier the higher-free energy antibonding orbital, just every bit they fill lower-energy diminutive orbitals earlier they fill higher-energy diminutive orbitals.

You lot can sentry animations visualizing the calculated atomic orbitals combining to form various molecular orbitals at the Orbitron website.
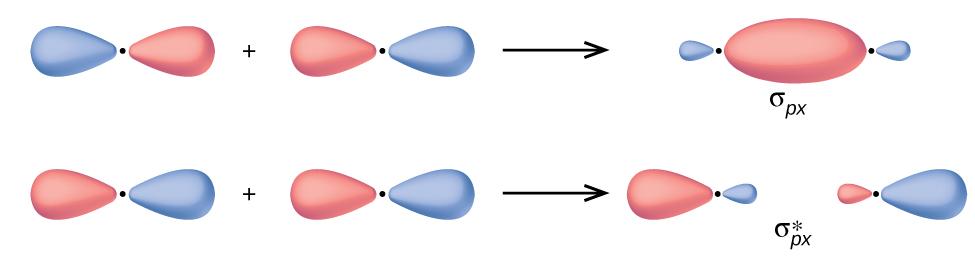
In p orbitals, the wave role gives rise to two lobes with opposite phases, coordinating to how a two-dimensional wave has both parts higher up and below the average. Nosotros betoken the phases by shading the orbital lobes unlike colors. When orbital lobes of the aforementioned phase overlap, constructive moving ridge interference increases the electron density. When regions of opposite phase overlap, the destructive wave interference decreases electron density and creates nodes. When p orbitals overlap end to cease, they create σ and σ* orbitals (Figure 4). If two atoms are located along the x-centrality in a Cartesian coordinate organization, the ii px orbitals overlap end to end and course σ px (bonding) and [latex]\sigma^*_{px}[/latex] (antibonding) (read as "sigma-p-x" and "sigma-p-x star," respectively). Simply equally with south-orbital overlap, the asterisk indicates the orbital with a node between the nuclei, which is a college-energy, antibonding orbital.
The side-by-side overlap of two p orbitals gives rise to a pi (π) bonding molecular orbital and a π* antibonding molecular orbital, as shown in Figure v. In valence bond theory, we describe π bonds as containing a nodal plane containing the internuclear axis and perpendicular to the lobes of the p orbitals, with electron density on either side of the node. In molecular orbital theory, we describe the π orbital by this same shape, and a π bond exists when this orbital contains electrons. Electrons in this orbital interact with both nuclei and assistance hold the two atoms together, making it a bonding orbital. For the out-of-phase combination, at that place are two nodal planes created, one along the internuclear axis and a perpendicular ane betwixt the nuclei.
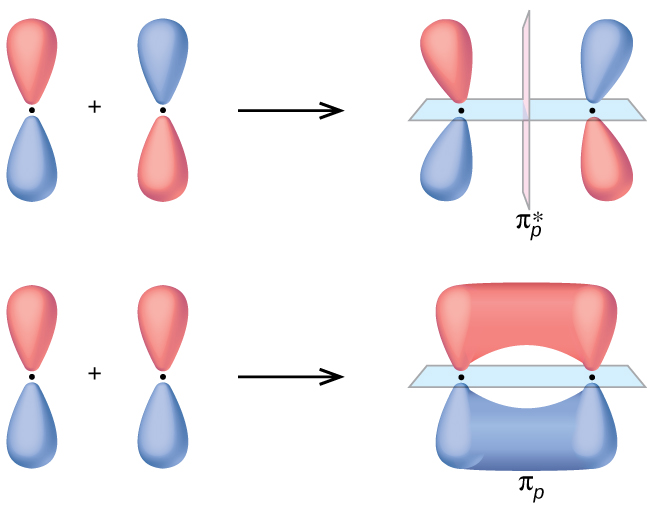
In the molecular orbitals of diatomic molecules, each atom also has 2 sets of p orbitals oriented next (py and pz ), and so these 4 diminutive orbitals combine pairwise to create ii π orbitals and ii π* orbitals. The π py and [latex]\pi^*_{py}[/latex] orbitals are oriented at right angles to the π pz and [latex]\pi^*_{pz}[/latex] orbitals. Except for their orientation, the π py and π pz orbitals are identical and have the same energy; they are degenerate orbitals. The [latex]\pi^*_{py}[/latex] and [latex]\pi^*_{px}[/latex] antibonding orbitals are too degenerate and identical except for their orientation. A total of six molecular orbitals results from the combination of the six atomic p orbitals in 2 atoms: σ px and [latex]\sigma^*_{px}[/latex], π py and [latex]\pi^*_{py}[/latex], π pz and [latex]\pi^*_{pz}[/latex].
Example 1
Molecular Orbitals
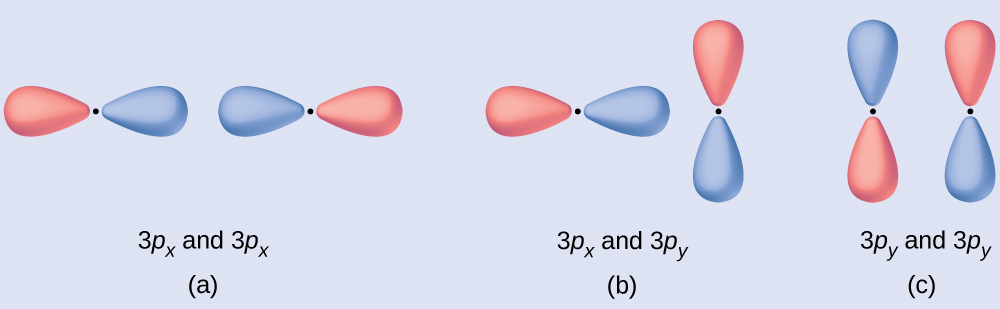
Predict what type (if any) of molecular orbital would result from adding the wave functions so each pair of orbitals shown overlap. The orbitals are all similar in energy.
Solution
(a) is an in-phase combination, resulting in a σthreep orbital
(b) will non result in a new orbital because the in-stage component (bottom) and out-of-stage component (pinnacle) abolish out. But orbitals with the correct alignment can combine.
(c) is an out-of-phase combination, resulting in a [latex]\pi^*_{3p}[/latex] orbital.
Bank check Your Learning
Label the molecular orbital shown as σ or π, bonding or antibonding and indicate where the node occurs.

Answer:
The orbital is located along the internuclear centrality, so information technology is a σ orbital. There is a node bisecting the internuclear axis, so it is an antibonding orbital.

Walter Kohn: Nobel Laureate
Walter Kohn (Figure vi) is a theoretical physicist who studies the electronic structure of solids. His piece of work combines the principles of quantum mechanics with advanced mathematical techniques. This technique, chosen density functional theory, makes it possible to compute properties of molecular orbitals, including their shape and energies. Kohn and mathematician John Pople were awarded the Nobel Prize in Chemical science in 1998 for their contributions to our agreement of electronic structure. Kohn likewise made significant contributions to the physics of semiconductors.

Kohn's biography has been remarkable outside the realm of physical chemistry as well. He was born in Austria, and during World War II he was part of the Kindertransport plan that rescued 10,000 children from the Nazi regime. His summertime jobs included discovering gilt deposits in Canada and helping Polaroid explicate how its instant film worked. Although he is at present an emeritus professor, he is still actively working on projects involving global warming and renewable energy.
Computational Chemistry in Drug Design
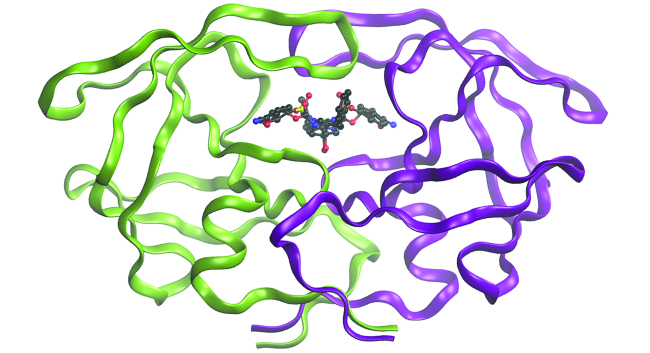
While the descriptions of bonding described in this chapter involve many theoretical concepts, they also have many practical, existent-globe applications. For instance, drug design is an important field that uses our understanding of chemical bonding to develop pharmaceuticals. This interdisciplinary area of study uses biology (understanding diseases and how they operate) to identify specific targets, such every bit a binding site that is involved in a disease pathway. By modeling the structures of the binding site and potential drugs, computational chemists can predict which structures can fit together and how effectively they will bind (meet Effigy vii). Thousands of potential candidates can be narrowed down to a few of the most promising candidates. These candidate molecules are then carefully tested to decide side furnishings, how effectively they can exist transported through the trunk, and other factors. Dozens of important new pharmaceuticals take been discovered with the assist of computational chemistry, and new research projects are underway.
Molecular Orbital Free energy Diagrams
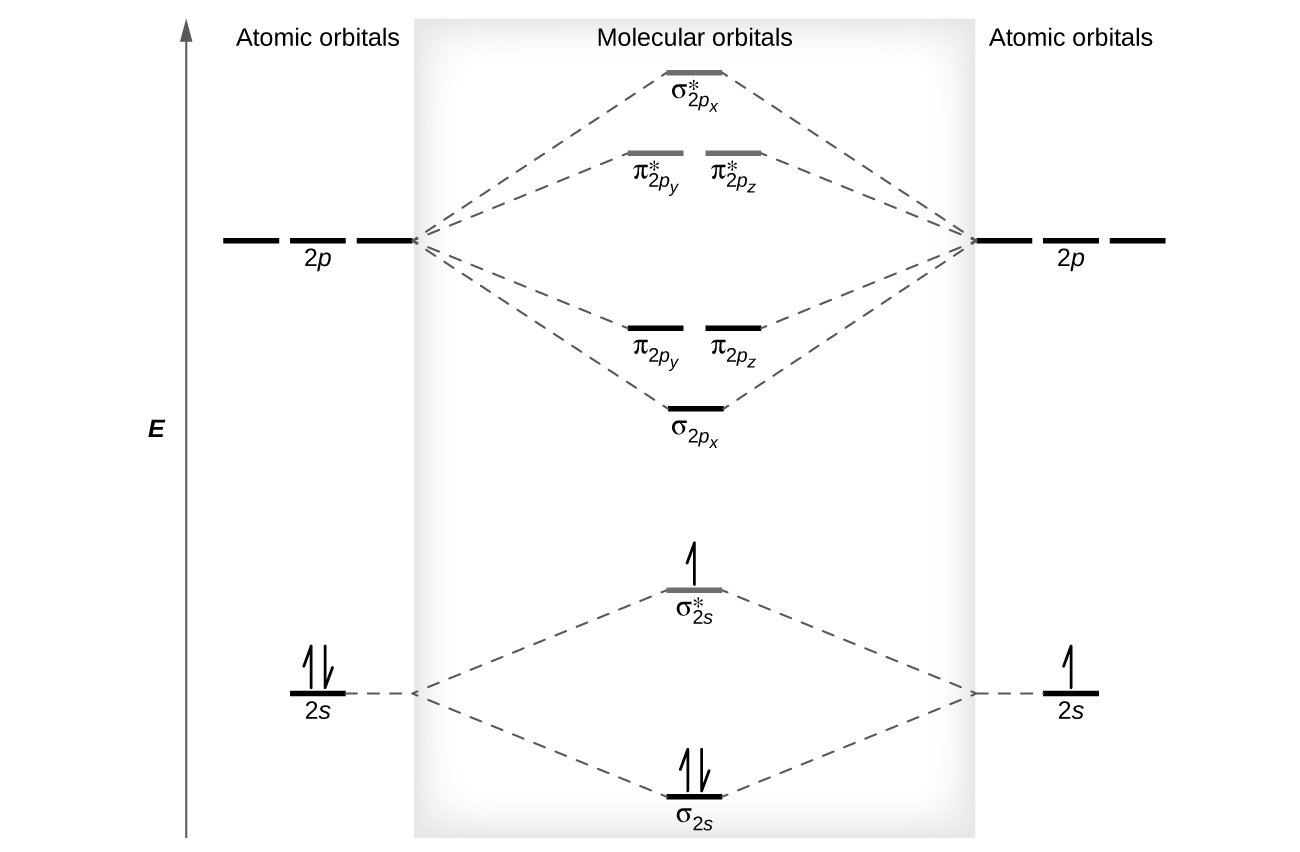
The relative energy levels of atomic and molecular orbitals are typically shown in a molecular orbital diagram (Figure 8). For a diatomic molecule, the diminutive orbitals of ane cantlet are shown on the left, and those of the other atom are shown on the right. Each horizontal line represents one orbital that can hold ii electrons. The molecular orbitals formed by the combination of the atomic orbitals are shown in the center. Dashed lines bear witness which of the atomic orbitals combine to form the molecular orbitals. For each pair of diminutive orbitals that combine, one lower-energy (bonding) molecular orbital and ane higher-energy (antibonding) orbital result. Thus we can run into that combining the six 2p atomic orbitals results in three bonding orbitals (one σ and 2 π) and iii antibonding orbitals (one σ* and ii π*).
We predict the distribution of electrons in these molecular orbitals by filling the orbitals in the same way that we fill up atomic orbitals, by the Aufbau principle. Lower-energy orbitals fill get-go, electrons spread out among degenerate orbitals before pairing, and each orbital can concord a maximum of two electrons with opposite spins (Effigy eight). Just as we write electron configurations for atoms, nosotros tin can write the molecular electronic configuration by listing the orbitals with superscripts indicating the number of electrons present. For clarity, nosotros place parentheses effectually molecular orbitals with the same energy. In this case, each orbital is at a different free energy, so parentheses separate each orbital. Thus we would expect a diatomic molecule or ion containing seven electrons (such as Beii +) would have the molecular electron configuration [latex](\sigma_{1s})^two[/latex] [latex](\sigma^*_{1s})^2[/latex] [latex](\sigma_{2s})^2[/latex] [latex](\sigma^*_{2s})^1[/latex]. It is mutual to omit the core electrons from molecular orbital diagrams and configurations and include simply the valence electrons.
Bond Lodge
The filled molecular orbital diagram shows the number of electrons in both bonding and antibonding molecular orbitals. The net contribution of the electrons to the bond strength of a molecule is identified past determining the bond order that results from the filling of the molecular orbitals past electrons.
When using Lewis structures to depict the distribution of electrons in molecules, we ascertain bail order as the number of bonding pairs of electrons between 2 atoms. Thus a single bond has a bond order of 1, a double bail has a bond order of ii, and a triple bail has a bail order of 3. We define bail society differently when we utilise the molecular orbital clarification of the distribution of electrons, only the resulting bond guild is usually the same. The MO technique is more than accurate and tin can handle cases when the Lewis structure method fails, merely both methods describe the same phenomenon.
In the molecular orbital model, an electron contributes to a bonding interaction if it occupies a bonding orbital and information technology contributes to an antibonding interaction if information technology occupies an antibonding orbital. The bond order is calculated by subtracting the destabilizing (antibonding) electrons from the stabilizing (bonding) electrons. Since a bond consists of two electrons, we split past two to become the bond order. We tin can determine bail gild with the post-obit equation:
[latex]\text{bail order} = \frac{(\text{number of bonding electrons}) - (\text{number of antibonding electrons})}{2}[/latex]
The guild of a covalent bail is a guide to its force; a bail betwixt two given atoms becomes stronger as the bond gild increases (Table 1 in Chapter 8.1 Valence Bail Theory). If the distribution of electrons in the molecular orbitals between two atoms is such that the resulting bond would take a bond order of null, a stable bond does not form. We next look at some specific examples of MO diagrams and bond orders.
Bonding in Diatomic Molecules
A dihydrogen molecule (Hii) forms from two hydrogen atoms. When the atomic orbitals of the ii atoms combine, the electrons occupy the molecular orbital of everyman energy, the σ1s bonding orbital. A dihydrogen molecule, Hii, readily forms because the energy of a H2 molecule is lower than that of two H atoms. The σ1southward orbital that contains both electrons is lower in free energy than either of the 2 1s atomic orbitals.
A molecular orbital can concord two electrons, and so both electrons in the H2 molecule are in the σ1south bonding orbital; the electron configuration is [latex](\sigma_{1s})^2[/latex]. Nosotros represent this configuration by a molecular orbital energy diagram (Figure 9) in which a single up pointer indicates i electron in an orbital, and ii (upward and downward) arrows indicate ii electrons of reverse spin.
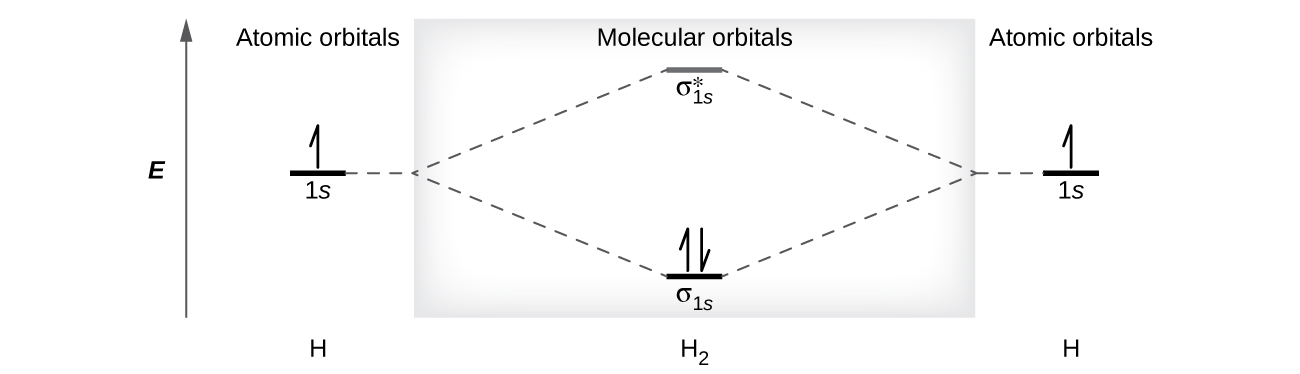
A dihydrogen molecule contains two bonding electrons and no antibonding electrons and then nosotros take
[latex]\text{bail gild in H}_2 = \frac{(2 - 0)}{2} = 1[/latex]
Because the bond gild for the H–H bond is equal to 1, the bond is a single bond.
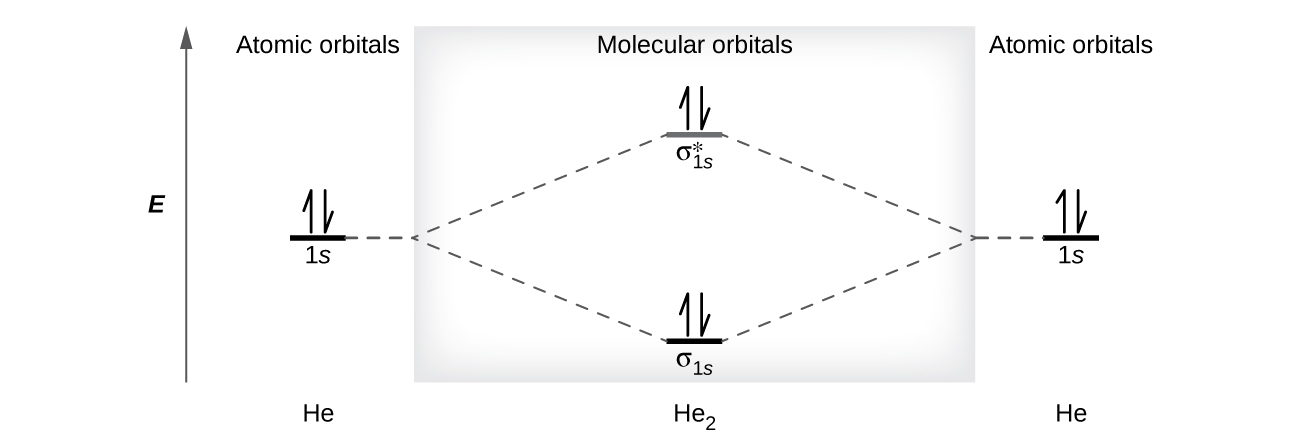
A helium atom has two electrons, both of which are in its 1south orbital. Two helium atoms do not combine to form a dihelium molecule, Hetwo, with iv electrons, because the stabilizing effect of the 2 electrons in the lower-energy bonding orbital would be offset past the destabilizing result of the 2 electrons in the higher-energy antibonding molecular orbital. We would write the hypothetical electron configuration of He2 as [latex](\sigma_{1s})^2[/latex] [latex](\sigma^*_{1s})^2[/latex] as in Figure 10. The net free energy change would exist nil, so there is no driving force for helium atoms to course the diatomic molecule. In fact, helium exists as discrete atoms rather than every bit diatomic molecules. The bond society in a hypothetical dihelium molecule would be zero.
[latex]\text{bail social club in He}_2 = \frac{(2 - two)}{2} = 0[/latex]
A bond order of zero indicates that no bond is formed between two atoms.
The Diatomic Molecules of the Second Period
Eight possible homonuclear diatomic molecules might exist formed by the atoms of the second period of the periodic table: Litwo, Be2, Bii, Ctwo, N2, Oii, Fii, and Ne2. However, we can predict that the Beii molecule and the Ne2 molecule would not be stable. Nosotros can see this by a consideration of the molecular electron configurations (Tabular array 3).
We predict valence molecular orbital electron configurations only equally we predict electron configurations of atoms. Valence electrons are assigned to valence molecular orbitals with the lowest possible energies. Consistent with Hund's rule, whenever there are ii or more than degenerate molecular orbitals, electrons fill each orbital of that type singly earlier any pairing of electrons takes place.
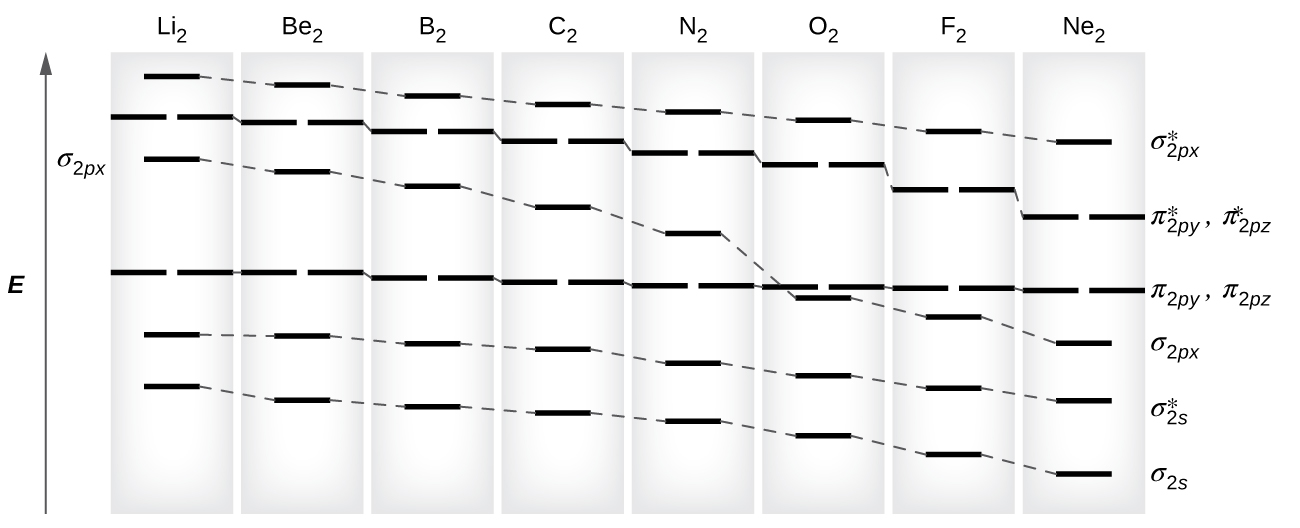
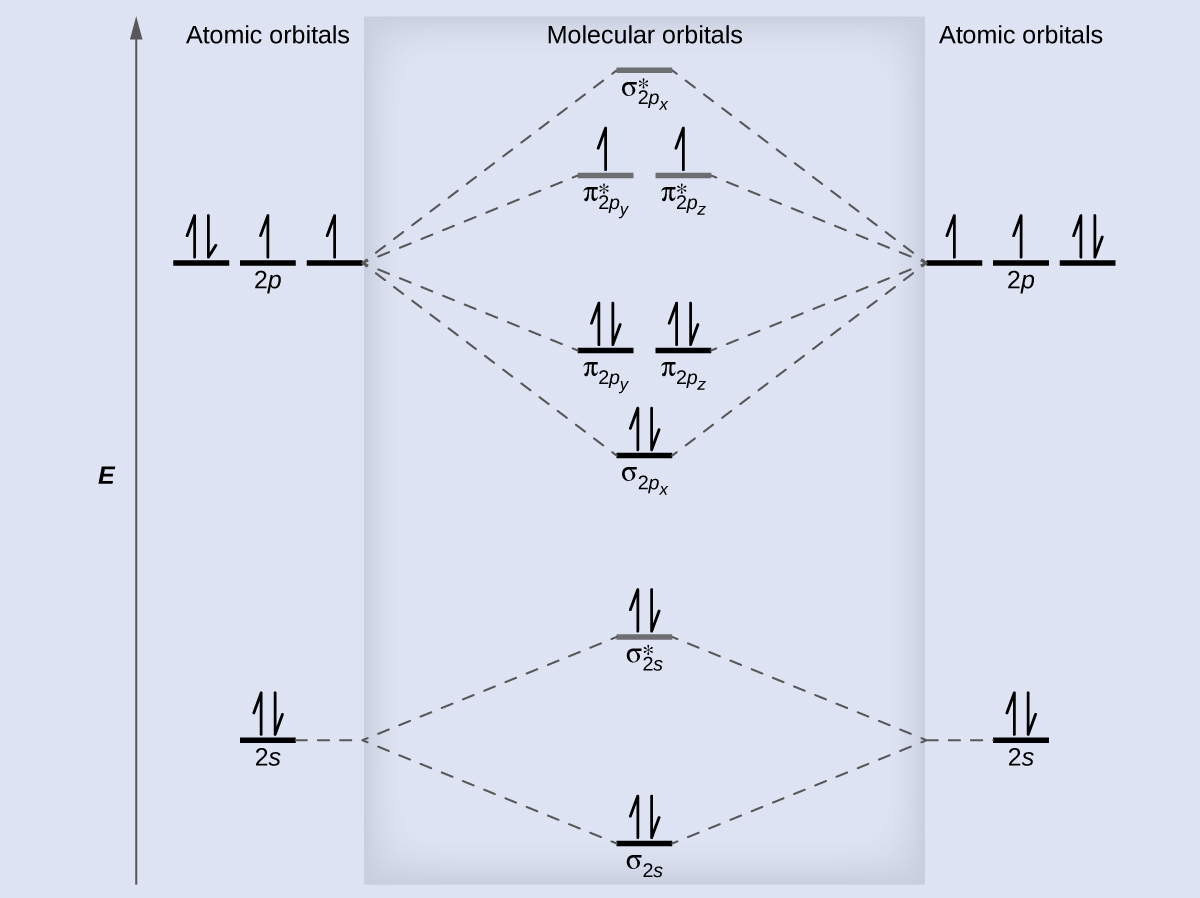
As nosotros saw in valence bond theory, σ bonds are generally more than stable than π bonds formed from degenerate atomic orbitals. Similarly, in molecular orbital theory, σ orbitals are usually more than stable than π orbitals. Nevertheless, this is non e'er the case. The MOs for the valence orbitals of the second period are shown in Figure 11. Looking at Netwo molecular orbitals, we meet that the order is consistent with the generic diagram shown in the previous section. Yet, for atoms with iii or fewer electrons in the p orbitals (Li through N) we find a dissimilar pattern, in which the σ p orbital is college in free energy than the π p set. Obtain the molecular orbital diagram for a homonuclear diatomic ion by adding or subtracting electrons from the diagram for the neutral molecule.

You tin exercise labeling and filling molecular orbitals with this interactive tutorial from the University of Sydney.
This switch in orbital ordering occurs because of a phenomenon called southward-p mixing. s-p mixing does non create new orbitals; information technology just influences the energies of the existing molecular orbitals. The σs wavefunction mathematically combines with the σp wavefunction, with the issue that the σsouthward orbital becomes more than stable, and the σp orbital becomes less stable (Figure 12). Similarly, the antibonding orbitals too undergo s-p mixing, with the σs* condign more stable and the σp* becoming less stable.
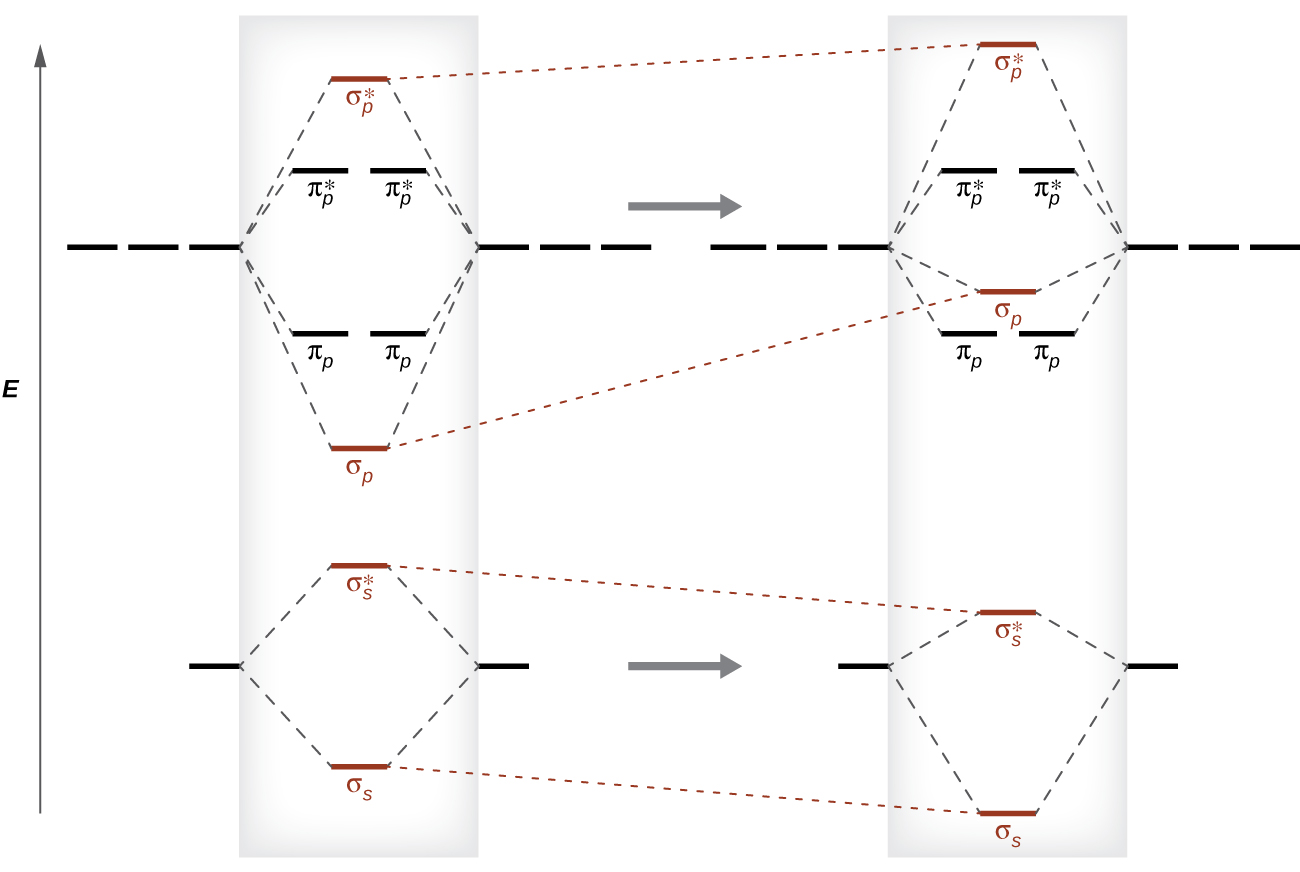
s-p mixing occurs when the southward and p orbitals have similar energies. When a single p orbital contains a pair of electrons, the human action of pairing the electrons raises the free energy of the orbital. Thus the 2p orbitals for O, F, and Ne are higher in energy than the 2p orbitals for Li, Be, B, C, and N. Because of this, O2, F2, and Northward2 only accept negligible s-p mixing (non sufficient to change the energy ordering), and their MO diagrams follow the normal pattern, as shown in Figure 11. All of the other period two diatomic molecules do have southward-p mixing, which leads to the pattern where the σp orbital is raised higher up the πp set.
Using the MO diagrams shown in Effigy eleven, nosotros can add in the electrons and make up one's mind the molecular electron configuration and bail guild for each of the diatomic molecules. As shown in Tabular array three, Exist2 and Neii molecules would have a bail order of 0, and these molecules do non exist.
| Molecule | Electron Configuration | Bond Lodge |
|---|---|---|
| Li2 | [latex](\sigma_{2s})^two[/latex] | 1 |
| Betwo (unstable) | [latex](\sigma_{2s})^2 (\sigma^*_{2s})^2[/latex] | 0 |
| Bii | [latex](\sigma_{2s})^ii (\sigma^*_{2s})^2 (\pi_{2py}, \pi_{2pz})^2[/latex] | 1 |
| Ctwo | [latex](\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2py}, \pi_{2pz})^iv[/latex] | 2 |
| N2 | [latex](\sigma_{2s})^2 (\sigma^*_{2s})^ii (\pi_{2py}, \pi_{2pz})^4 (\sigma_{2px})^2[/latex] | 3 |
| O2 | [latex](\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2px})^2 (\pi_{2py}, \pi_{2pz})^4 (\pi^*_{2py}, \pi^*_{2pz})^2[/latex] | 2 |
| F2 | [latex](\sigma_{2s})^two (\sigma^*_{2s})^two (\sigma_{2px})^2 (\pi_{2py}, \pi_{2pz})^iv (\pi^*_{2py}, \pi^*_{2pz})^4[/latex] | 1 |
| Neii (unstable) | [latex](\sigma_{2s})^ii (\sigma^*_{2s})^2 (\sigma_{2px})^two (\pi_{2py}, \pi_{2pz})^4 (\pi^*_{2py}, \pi^*_{2pz})^four (\sigma^*_{2px})^2[/latex] | 0 |
| Tabular array three. Electron Configuration and Bond Order for Molecular Orbitals in Homonuclear Diatomic Molecules of Flow Two Elements | ||
The combination of two lithium atoms to form a lithium molecule, Li2, is analogous to the formation of Htwo, just the atomic orbitals involved are the valence 2due south orbitals. Each of the 2 lithium atoms has ane valence electron. Hence, we have two valence electrons available for the σ2south bonding molecular orbital. Because both valence electrons would be in a bonding orbital, we would predict the Li2 molecule to be stable. The molecule is, in fact, present in appreciable concentration in lithium vapor at temperatures near the boiling point of the chemical element. All of the other molecules in Table iii with a bail guild greater than null are also known.
The O2 molecule has enough electrons to half fill the ([latex]\pi^*_{2py}[/latex], [latex]\pi^*_{2pz}[/latex]) level. We await the two electrons that occupy these two degenerate orbitals to be unpaired, and this molecular electronic configuration for O2 is in accord with the fact that the oxygen molecule has two unpaired electrons (Figure xiv). The presence of 2 unpaired electrons has proved to be difficult to explain using Lewis structures, only the molecular orbital theory explains it quite well. In fact, the unpaired electrons of the oxygen molecule provide a strong piece of support for the molecular orbital theory.
Band Theory
When two identical atomic orbitals on different atoms combine, two molecular orbitals outcome (run across Figure three). The bonding orbital is lower in energy than the original diminutive orbitals because the diminutive orbitals are in-phase in the molecular orbital. The antibonding orbital is higher in energy than the original diminutive orbitals because the atomic orbitals are out-of-phase.
In a solid, similar things happen, only on a much larger scale. Remember that fifty-fifty in a small sample there are a huge number of atoms (typically > 1023 atoms), and therefore a huge number of atomic orbitals that may exist combined into molecular orbitals. When N valence diminutive orbitals, all of the same energy and each containing one (ane) electron, are combined, N/2 (filled) bonding orbitals and Northward/ii (empty) antibonding orbitals will result. Each bonding orbital will show an energy lowering every bit the atomic orbitals are mostly in-stage, simply each of the bonding orbitals will be a little different and have slightly different energies. The antibonding orbitals will show an increase in free energy equally the atomic orbitals are more often than not out-of-phase, only each of the antibonding orbitals volition also be a little unlike and take slightly different energies. The allowed energy levels for all the bonding orbitals are and then shut together that they form a band, called the valence band. Besides, all the antibonding orbitals are very close together and form a band, called the conduction band. Figure 13 shows the bands for three important classes of materials: insulators, semiconductors, and conductors.
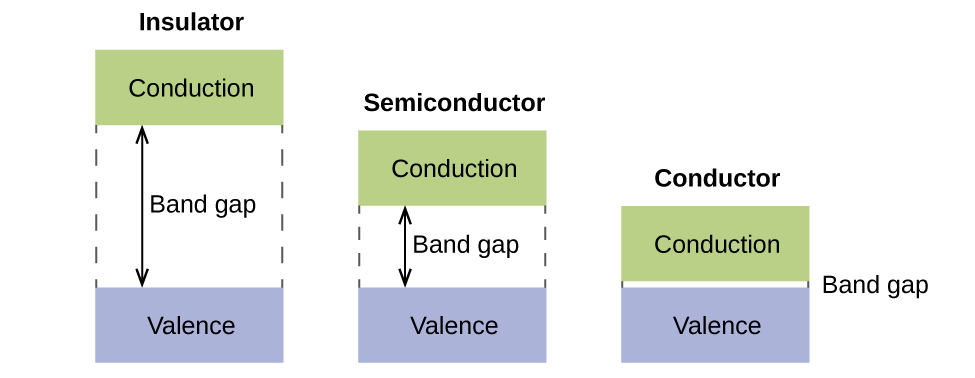
In social club to conduct electricity, electrons must move from the filled valence band to the empty conduction band where they tin can motion throughout the solid. The size of the ring gap, or the energy divergence between the summit of the valence band and the bottom of the conduction band, determines how easy it is to movement electrons between the bands. Only a small amount of energy is required in a conductor because the ring gap is very pocket-sized. This small free energy difference is "easy" to overcome, then they are good conductors of electricity. In an insulator, the band gap is so "large" that very few electrons motion into the conduction ring; as a result, insulators are poor conductors of electricity. Semiconductors conduct electricity when "moderate" amounts of energy are provided to motion electrons out of the valence band and into the conduction band. Semiconductors, such as silicon, are plant in many electronics.
Semiconductors are used in devices such as computers, smartphones, and solar cells. Solar cells produce electricity when lite provides the free energy to move electrons out of the valence ring. The electricity that is generated may then exist used to power a calorie-free or tool, or it can be stored for subsequently use by charging a battery. Equally of December 2014, up to 46% of the free energy in sunlight could be converted into electricity using solar cells.
Example 2
Molecular Orbital Diagrams, Bail Order, and Number of Unpaired Electrons
Draw the molecular orbital diagram for the oxygen molecule, O2. From this diagram, calculate the bond gild for Oii. How does this diagram business relationship for the paramagnetism of O2?
Solution
We draw a molecular orbital free energy diagram similar to that shown in Figure eleven. Each oxygen atom contributes half-dozen electrons, then the diagram appears as shown in Figure 14.
Nosotros calculate the bond society as
[latex]\text{O}_2 = \frac{8 - 4}{two} = 2[/latex]
Oxygen's paramagnetism is explained by the presence of two unpaired electrons in the (π2py , π2pz )* molecular orbitals.
Bank check Your Learning
The chief component of air is North2. From the molecular orbital diagram of Due north2, predict its bail guild and whether it is diamagnetic or paramagnetic.
Answer:
N2 has a bond society of 3 and is diamagnetic.
Example 3
Ion Predictions with MO Diagrams
Give the molecular orbital configuration for the valence electrons in C2 two−. Will this ion exist stable?
Solution
Looking at the appropriate MO diagram, we run into that the π orbitals are lower in energy than the σ p orbital. The valence electron configuration for C2 is [latex](\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2py}, \pi_{2pz})^four[/latex]. Adding ii more than electrons to generate the Cii 2− anion will requite a valence electron configuration of [latex](\sigma_{2s})^ii (\sigma^*_{2s})^2 (\pi_{2py}, \pi_{2pz})^four (\sigma_{2px})^2[/latex]. Since this has six more bonding electrons than antibonding, the bond club will be 3, and the ion should be stable.
Check Your Learning
How many unpaired electrons would be present on a Be2 2− ion? Would it exist paramagnetic or diamagnetic?
Reply:
two, paramagnetic

Creating molecular orbital diagrams for molecules with more than than two atoms relies on the aforementioned basic ideas as the diatomic examples presented hither. However, with more atoms, computers are required to calculate how the diminutive orbitals combine. Run across three-dimensional drawings of the molecular orbitals for C6H6.
Key Concepts and Summary
Molecular orbital (MO) theory describes the behavior of electrons in a molecule in terms of combinations of the atomic moving ridge functions. The resulting molecular orbitals may extend over all the atoms in the molecule. Bonding molecular orbitals are formed by in-stage combinations of diminutive wave functions, and electrons in these orbitals stabilize a molecule. Antibonding molecular orbitals result from out-of-phase combinations of atomic wave functions and electrons in these orbitals brand a molecule less stable. Molecular orbitals located along an internuclear axis are called σ MOs. They can be formed from due south orbitals or from p orbitals oriented in an stop-to-end fashion. Molecular orbitals formed from p orbitals oriented in a side-by-side fashion have electron density on contrary sides of the internuclear axis and are chosen π orbitals.
We can depict the electronic construction of diatomic molecules by applying molecular orbital theory to the valence electrons of the atoms. Electrons fill molecular orbitals following the same rules that apply to filling atomic orbitals; Hund'due south rule and the Aufbau principle tell us that lower-energy orbitals will fill up first, electrons will spread out earlier they pair up, and each orbital tin concord a maximum of 2 electrons with contrary spins. Materials with unpaired electrons are paramagnetic and attracted to a magnetic field, while those with all-paired electrons are diamagnetic and repelled by a magnetic field. Correctly predicting the magnetic backdrop of molecules is in advantage of molecular orbital theory over Lewis structures and valence bond theory.
Key Equations
- [latex]\text{bail order} = \frac{(\text{number of bonding electron}) - (\text{number of antibonding electrons})}{ii}[/latex]
Chemical science End of Affiliate Exercises
- Sketch the distribution of electron density in the bonding and antibonding molecular orbitals formed from two south orbitals and from ii p orbitals.
- How are the following similar, and how do they differ?
(a) σ molecular orbitals and π molecular orbitals
(b) ψ for an atomic orbital and ψ for a molecular orbital
(c) bonding orbitals and antibonding orbitals
- If molecular orbitals are created by combining five atomic orbitals from atom A and five diminutive orbitals from atom B combine, how many molecular orbitals will consequence?
- Can a molecule with an odd number of electrons always be diamagnetic? Explain why or why not.
- Can a molecule with an even number of electrons e'er be paramagnetic? Explain why or why not.
- Why are bonding molecular orbitals lower in energy than the parent atomic orbitals?
- Calculate the bond gild for an ion with this configuration:
[latex](\sigma_{2s})^2 (\sigma^*_{2s})^two (\sigma_{2px})^ii (\pi_{2py} , \pi_{2pz})^four (\pi^*_{2py} , \pi^*_{2pz})^3[/latex]
- Explain why an electron in the bonding molecular orbital in the Htwo molecule has a lower energy than an electron in the onedue south atomic orbital of either of the separated hydrogen atoms.
- Predict the valence electron molecular orbital configurations for the following, and state whether they will exist stable or unstable ions.
(a) Na2 two+
(b) Mg2 2+
(c) Al2 2+
(d) Si2 ii+
(eastward) P2 2+
(f) Southwardtwo 2+
(g) F2 2+
(h) Artwo 2+
- Determine the bond society of each member of the post-obit groups, and determine which member of each group is predicted by the molecular orbital model to take the strongest bond.
(a) Hii, Htwo +, H2 −
(b) O2, O2 two+, O2 2−
(c) Litwo, Be2 +, Be2
(d) F2, Ftwo +, Ftwo −
(due east) North2, N2 +, Northward2 −
- For the showtime ionization energy for an N2 molecule, what molecular orbital is the electron removed from?
- Compare the diminutive and molecular orbital diagrams to place the fellow member of each of the following pairs that has the highest beginning ionization energy (the well-nigh tightly leap electron) in the gas phase:
(a) H and Htwo
(b) N and N2
(c) O and Oii
(d) C and C2
(e) B and B2
- Which of the period 2 homonuclear diatomic molecules are predicted to be paramagnetic?
- A friend tells you that the 2s orbital for fluorine starts off at a much lower energy than the 2southward orbital for lithium, and so the resulting σ2south molecular orbital in F2 is more stable than in Liii. Do you lot agree?
- True or false: Boron contains 2southward 22p i valence electrons, then just one p orbital is needed to form molecular orbitals.
- What accuse would be needed on F2 to generate an ion with a bond club of 2?
- Predict whether the MO diagram for S2 would show s-p mixing or not.
- Explain why N2 2+ is diamagnetic, while O2 four+, which has the same number of valence electrons, is paramagnetic.
- Using the MO diagrams, predict the bond gild for the stronger bond in each pair:
(a) B2 or B2 +
(b) F2 or F2 +
(c) O2 or O2 2+
(d) Ctwo + or C2 −
Glossary
- antibonding orbital
- molecular orbital located outside of the region between two nuclei; electrons in an antibonding orbital destabilize the molecule
- bond order
- number of pairs of electrons betwixt two atoms; it tin can be establish by the number of bonds in a Lewis construction or by the difference between the number of bonding and antibonding electrons divided by two
- bonding orbital
- molecular orbital located between two nuclei; electrons in a bonding orbital stabilize a molecule
- degenerate orbitals
- orbitals that take the same energy
- diamagnetism
- phenomenon in which a material is non magnetic itself only is repelled past a magnetic field; it occurs when in that location are only paired electrons present
- homonuclear diatomic molecule
- molecule consisting of ii identical atoms
- linear combination of diminutive orbitals
- technique for combining atomic orbitals to create molecular orbitals
- molecular orbital
- region of infinite in which an electron has a high probability of being found in a molecule
- molecular orbital diagram
- visual representation of the relative free energy levels of molecular orbitals
- molecular orbital theory
- model that describes the behavior of electrons delocalized throughout a molecule in terms of the combination of atomic wave functions
- paramagnetism
- phenomenon in which a cloth is non magnetic itself merely is attracted to a magnetic field; it occurs when there are unpaired electrons present
- π bonding orbital
- molecular orbital formed by side-by-side overlap of atomic orbitals, in which the electron density is plant on opposite sides of the internuclear axis
- π* bonding orbital
- antibonding molecular orbital formed by out of phase side-by-side overlap of diminutive orbitals, in which the electron density is found on both sides of the internuclear axis, and there is a node between the nuclei
- σ bonding orbital
- molecular orbital in which the electron density is found along the axis of the bond
- σ* bonding orbital
- antibonding molecular orbital formed by out-of-phase overlap of atomic orbital along the centrality of the bond, generating a node betwixt the nuclei
- south-p mixing
- change that causes σ p orbitals to be less stable than π p orbitals due to the mixing of due south and p-based molecular orbitals of similar energies.
Solutions
Answers to Chemical science End of Affiliate Exercises
ii. (a) Similarities: Both are bonding orbitals that can contain a maximum of two electrons. Differences: σ orbitals are stop-to-finish combinations of atomic orbitals, whereas π orbitals are formed by side-by-side overlap of orbitals. (b) Similarities: Both are quantum-mechanical constructs that stand for the probability of finding the electron near the atom or the molecule. Differences: ψ for an atomic orbital describes the behavior of only one electron at a time based on the cantlet. For a molecule, ψ represents a mathematical combination of diminutive orbitals. (c) Similarities: Both are orbitals that tin contain two electrons. Differences: Bonding orbitals issue in property two or more than atoms together. Antibonding orbitals have the effect of destabilizing any bonding that has occurred.
four. An odd number of electrons tin never be paired, regardless of the arrangement of the molecular orbitals. It will always be paramagnetic.
6. Bonding orbitals take electron density in close proximity to more than ane nucleus. The interaction between the bonding positively charged nuclei and negatively charged electrons stabilizes the system.
eight. The pairing of the ii bonding electrons lowers the energy of the arrangement relative to the energy of the nonbonded electrons.
x. (a) Htwo bond lodge = 1, H2 + bail order = 0.v, H2 − bond order = 0.v, strongest bond is Htwo; (b) Oii bail guild = 2, Oii 2+ bail social club = 3; O2 2− bond society = one, strongest bond is O2 2+; (c) Li2 bond gild = ane, Exist2 + bail order = 0.5, Beii bond order = 0, strongest bond is Li2;(d) F2 bond gild = 1, F2 + bond society = 1.5, F2 − bail order = 0.5, strongest bond is Ftwo +; (e) Nii bail social club = 3, N2 + bond society = 2.v, Nii − bond order = 2.5, strongest bail is N2
12. (a) Htwo; (b) Northward2; (c) O; (d) Ctwo; (e) B2
14. Yes, fluorine is a smaller atom than Li, so atoms in the iisouth orbital are closer to the nucleus and more stable.
16. ii+
18. N2 has s-p mixing, so the π orbitals are the last filled in N2 ii+. O2 does not have south-p mixing, so the σ p orbital fills before the π orbitals.
Source: https://opentextbc.ca/chemistry/chapter/8-4-molecular-orbital-theory/
0 Response to "How to Draw Molecular Orbital Diagram of Co"
Post a Comment